| When John Gurdon, the Nobel prize-winning developmental cell biologist, was asked to choose his favourite scientific study as the subject of an essay for the journal Cell, he surprised many by choosing a work about snails (Gurdon, 2005). The study in question (largely the work of amateur scientists, as if happens) was Boycott and Diver’s insightful description of shell-coiling dimorphism in the fresh-water species Lymnaea peregra (Diver, Boycott & Garstang, 1925; Boycott & Diver, 1927; Boycott et al.,1931). As almost every malacologist learns at their mother’s knee, most species of snail have shells that coil resolutely and invariably to the right (they are dextral), while a much smaller number of species favour the left (they are sinistral). A tiny handful of species, Lymnaea peregra amongst them, express both forms in large enough numbers for them to be called dimorphic. This otherwise obscure piece of snail natural history captured the imagination of John Gurdon because of what it tells us about the fundamental basis of animal development. Boycott and Diver’s breakthrough was to show that the coil direction of Lymnaea was under the control of a single gene with its main activity during the formation of the egg in the mother before fertilisation (Boycott & Diver, 1927). This same gene is active in the early development of all animals helping to establish the left-right axis along which the organs of the body develop and as such it is one of the handful of most fundamental developmental genes so far discovered (Hierck et al. 2005).
Scientific interest in Boycott and Diver’s discovery extends far beyond developmental biology. In a series of theoretical studies in the 1980s and ‘90s, the evolutionary biologist and malacologist Edmund Gittenberger predicted two things about sinistral and dextral snails (Gittenberger, 1988; VanBattenburg & Gittenberger, 1996). Firstly, the switch of a few individuals to sinistral in an otherwise dextral species (or vice versa) could result in the origin of a new species. This is because mating between sinistral and dextral individuals is often impossible or at least difficult (Asami, Cowie & Ohbayashi, 1998). The reason for this lies in the positioning of the external connection to the genitalia on one side of the body close to the respiratory pore. Two snails with the same coil are able to align their genital pores correctly so that gametes can be exchanged. Two snails of opposite coil, on the other hand, can align themselves at best with difficulty. To Gittenberger this immediately suggested a mechanism for forming a new species. If coil direction could prevent parts of the same species reproducing with each other they would, using the generally accepted definition, instantly be separate species and be set on the path to becoming recognisably different. Not only would this origin be instantaneous, it would, as Boycott and Diver had shown, result from the expression of a single gene; something considered unlikely if not impossible in the scientific cannon of the time. Nearly two decades of investigation has shown Gittenberger’s idea of single-gene speciation to be a clear theoretical possibility but with some restrictions that make it less likely in reality (Orr, 1991; Stone & Björklund, 2002). But despite these complications, in 2003 Rei Ueshima and Hiro Asami finally published proof of speciation by shellcoil switching in the Japanese genus Euhadra (Ueshima & Asami, 2003). Gittenberger’s other idea concerned the maintenance of shell-coil dimorphism: his theoretical models showed it should not exist. Any population with both forms should become two species if sinistrals and dextrals cannot mate or revert to just dextrals or just sinistrals if they can. The one thing the models said definitely could not occur was long-term stability of the dimorphism. But Lymaea peregra and the other dimorphic species, though small in number, are incontrovertible proof that sinistral and dextral morphs can quite happily co-exist in the same species indefinitely. Something was clearly missing from the understanding of shell-coiling. In 2003, with a research grant from The Conchological Society, my colleague Menno Schilthuizen and I set about trying to find this missing factor in the shell-coil dimorphism story. We first needed a convenient group of study species. The South-East Asian tree snail subgenus Amphidromus s. str. (Camaemindae), with 28 dimorphic species out of a total of 36, was an obvious choice, not least because a number of those species were present in Borneo where Menno was then working at the Institute for Tropical Biology and Conservation in Kota Kinabalu (Sutcharit & Panha, 2006; Sutcharit, Asami & Panha, 2007). We focussed our efforts initially on Amphidromus martensi, a large, conspicuous and above all relatively common species of the rainforest canopy, and travelled to the Kinabtangan basin in the eastern part of Sabah, northern Borneo, to begin our fieldwork. We reasoned there might be two explanations for dimorphism. Up until that time, all collections of A.martensi had followed standard practice and recorded the sample location. But A.martensi is a tree-dwelling species; it is quite possible for individual trees or small groups of trees to harbour separate populations of the snail. If these populations had done what Edmund Gittenberger’s equations predicted and contained only sinistral or only dextral forms, we may see very different things depending on the scale of sampling. At a small, tree-by-tree scale we may see separate populations of just sinistral or just dextral snails. At a broader scale (the scale at which conchological samples are generally taken) this mosaic of small populations may merge into one with, apparently, a mix of sinistral and dextral forms giving the appearance of general dimorphism. For our second explanation we suggested there may be some process acting on the snails that changes as the relative frequency of sinistrals and dextrals changes. As sinistrals become more common in a population, this process favours the minority dextrals until they themselves become more common. Now the process favours the rarer sinsitrals so that they, in turn, become more common. By always favouring the rarer form, this process (referred to as frequency-dependent selection) prevents a population from being taken over by one or other of the two forms and can maintain dimorphism indefinitely. The most likely agent of this process was a predator. Some predators of snails are themselves asymmetric and so could act in the way described. For example, Southeast Asian snakes of the family Pareatinae are asymmetric in their dentition so as to make it easier to remove a dextral snail from its shell. Sinistral shells are dropped more often and so have more chance of surviving a snake attack (Hoso, Asami & Hori, 2007). If a predator such as a snake was responsible for maintaining the dimorphism we would expect to see this reflected in the spatial pattern of sinstrality and dextrality observed in A.martensi. To test whether either of these processes was involved, we needed a precise spatial map of where individual A.martensi were in the forest. Unlike some of the botanists of old who used trained monkeys to sample plants from the forest canopy, we were forced to sample shells of Amphidromus collected from the forest floor. But, with the help of Berjaya bin Elahan of the Kinabatangan Orangutan Conservation Project, by the end of our time in the field we had the exact location of enough shells to answer our question. The answer we got, however, was not one we were expecting. There was no structure in the distribution of sinistral and dextral morphs at any spatial scale; wherever you looked the ratio of sinistral to dextral snails was around 50:50 (Craze, bin Elahan & Schilthuizen 2006). Puzzled by our findings I returned to the UK to continue analysing the results while Menno took a well deserved holiday on the Malaysian island of Pulau Kapas. Almost as soon as he got there he found it covered with large numbers of another Amphidromus species, A.inversus. Unlike A.martensi, these snails were not in the forest canopy but closer to ground level on the tree trunks at a height were they could be observed directly. Menno set about recording the location of sinistral and dextral snails as well as noting the numbers of mating pairs of each type: sinistral-sinistral, dextral-dextral and sinistral-dextral (Schilthuizen et al. 2005). When we analysed the results we found there were more sinistral-dextral pairs than we would have expected by chance. It seemed an individual A.inversus preferred to mate with a snail coiled in the opposite direction to itself, a behaviour that was considered disadvantageous in most species due to the problem of aligning genital pores. If this was the case we had accidentally found the process needed for our second explanation of frequency-dependent selection. An individual of the less frequent morph would always be sought out for mating and would be likely to produce more offspring on average than an individual of the more common morph. The tables would turn as soon as the morph that was originally rare became more common – it might very well miss out in favour of the rarer form. We even had evidence for why sinistral-dextral might be the preferred pairing. Using a technique of snap-freezing I developed with Andrew Barr (Craze & Barr, 2002), Menno had collected mating pairs. His dissections of these showed that the spermatophore produced by an individual A.inversus is coiled in the same way as its shell. There is also an asymmetry in the female genitalia at the point where the oviduct attaches to the sperm receiving organ (SRO). In a sinistral individual the oviduct branches to the left, in a dextral individual it branches to the right. When two A.inversus of opposite coil mate, the tip of the spermatophore lines up directly with the oviduct whereas if both snails are of the same coil, the tip points out into the SRO. As the SRO has evolved to digest sperm, a snail mating with an individual of opposite coil is likely to fertilise more of its partner’s eggs rather than lose sperm to digestion. Incorporating these new findings into computer models showed that dimorphism is quite easy to achieve and is stable in the long term (Schilthuizen et al. 2007). Thanks to the Society’s research grant we may have uncovered at least one of the reasons why this conspicuous and intriguing polymorphism persists against the expectations of theory. While the techniques of computer simulation and spatial statistics may not have been available to Boycott and Diver, we hope they would have been pleased to see the prime role played by careful fieldwork in our study. We also hope they would have been heartened that the spirit of scientific curiosity that motivated them and so impressed John Gurdon was still very much alive. Asami, T. Cowie, R.H. Ohbayashi, K. (1998) Evolution of mirror images by sexually asymmetric mating behavior in hermaphroditic snails. American Naturalist, 152, 225 – 236. Boycott, A.E. & Diver, C. (1927) The origin of an albino mutation in Lymnaea peregra. Nature, 119, 9. Boycott, A.E., Diver, C., Garstang, S. & Turner, F.M. (1931) The inheritance of sinistrality in Lymnaea peregra (Mollusca, Pulmonata). Philisophical Transactions of the Royal Society Series B. 219, 51 – 131. Craze, P.G. & Barr, A.G. (2002) The use of electrical component freezing spray as a method of killing and preparing snails. Journal of Molluscan Studies, 68, 191 – 192. Craze, P.G., bin Elahan, B. & Schilthuizen, M. (2006) Opposite shell-coiling morphs of the tropical land snail Amphidromus martensi show no spatial-scale effects. Ecography, 29, 477 – 486. (See also Ecography, 29, 941). Diver, C., Boyott, A.E. & Garstang, S. (1925) The inheritance of inverse symmetry in Limnea peregra. Journal of Genetics, 19, 113 – 200. Gittenberger, E. (1988) Sympatric speciation in snails; a largely neglected model. Evolution, 42, 826 – 828. Gurdon, J.B. (2005) Sinistral snails and gentleman scientists. Cell, 123, 751 – 753. DOI: 10.1016/j.cell.2005.11.015 Hierck, B.P., Witte, B., Poelmann, R.E., Gittenberger-de Groot, A.C. & Gittenberger, E. (2005) Chirality in snails is determined by highly conserved asymmetry genes. Journal of Molluscan Studies, 71, 192 – 195. Hoso, M., Asami, T. & Hori, M. (2007) Right-handed snakes: convergent evolution of asymmetry for functional specialization. Biology Letters, 3, 169 – 172. DOI: 10.1098/rsbl.2006.0600 Orr, H.A. (1991) Is single gene speciation possible? Evolution, 45, 764 – 769. Schilthuizen, M. Scott, B.J., Cabanban, A.S. & Craze, P.G. (2005) Population structure and coil dimorphism in a tropical land snail. Heredity, 95, 216 – 220. Schilthuizen, M., Craze, P.G., Cabanban, A.S., Davison, A., Stone, J., Gittenberger, E. & Scott, B.J. (2007) Sexual selection maintains whole-body chiral dimorphism in snails. Journal of Evolutionary Biology, 20, 1941 – 1949. DOI: 10.1111/j.1420- 9101.2007.01370.x Stone, J. & Björklund, M. (2002) Delayed prezygotic isolating mechanisms: evolution with a twist. Proceedings of the Royal Society of London Series B. 269, 861 – 865. Sutcharit, C. & Panha, S. (2006) Taxonomic review of the tree snail Amphidromus Albers, 1850 (Pulmonata : Camaenidae) in Thailand and adjacent areas: Subgenus Amphidromus. Journal of Molluscan Studies, 72, 1 – 30. Sutcharit, C., Asami, T. & Panha, S. (2007) Evolution of whole-body enantiomorphy in the tree snail genus Amphidromus. Journal of Evolutionary Biology, 20, 661 – 672. Ueshima, R. & Asami, T. (2003) Single-gene speciation by leftright reversal - A land-snail species of polyphyletic origin results from chirality constraints on mating. Nature, 425, 679. VanBattenburg, F.H.D. & Gittenberger, E. (1996) Ease of fixation of a change in coiling: Computer experiments on chirality in snails. Heredity, 76, 278 – 286. |
Sinistral and dextral specimens of Amphidromus martensi collected from Kinabatangan, Borneo
|

Amphidromus inversus on Kapas island, Malaysia. (a) One dextral (top) and five sinistral adults (approximate shell height: 40 mm); (b) inter-chiral copulation between a dextral (left) and a sinistral (right) individual; (c) a freshly laid clutch of eggs; (d) a sinistral (left) and a dextral (right) adult shell; (e) a large number of apically crushed shells in a rat midden; (f) a recently hatched egg clutch, all sinistral (mean shell diameter: 3 mm). [Reproduced from Schilthuizen et al. 2007, with permission from M. Schilthuizen and Blackwell Publishing].
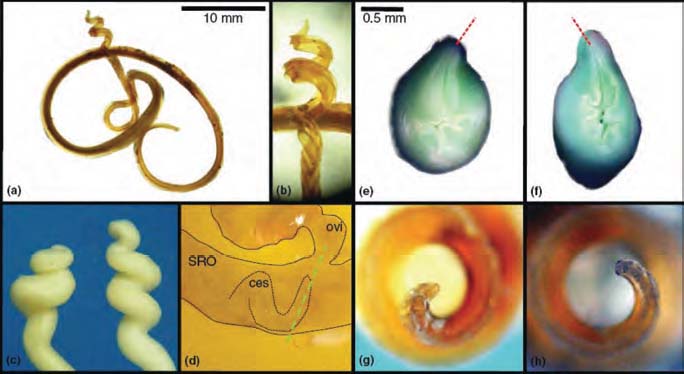
Amphidromus inversus reproductive anatomy. (a) An entire sinistral spermatophore; (b) the coiled expanded section (CES) of a sinistral spermatophore; (c) the epiphallic caeca (which produce the CES) of a sinistral (left) and a dextral (right) individual; (d) the CES of a recently deposited sinistral spermatophore at the junction of the spermatophore receiving organ (SRO) and the free oviduct (ovi) of a dextral individual (outlines added for clarity; the green line indicates the plane of the sections in e and f); (e, f) vaginal view of the entrance of the free oviduct in a dextral (e) and a sinistral (f) individual; the red arrows indicate the direction of attachment of the oviduct, which corresponds with the coil of the spermatophore tips (g, h) in an inter-chiral mating; (g, h) apical view of the CES in a sinistral (g) and a dextral (h) spermatophore. [Reproduced from Schilthuizen et al. 2007 with permission from M. Schilthuizen and Blackwell Publishing].


 Berjaya bin Elahan by the Kinabatangan River during a break in fieldwork
Berjaya bin Elahan by the Kinabatangan River during a break in fieldwork Menno Schilthuizen examining rock-dwelling snails
Menno Schilthuizen examining rock-dwelling snails