|
Introduction In terms of biodiversity, India is one of the richest and most highly endangered regions of the world. It is perhaps the only Asian country with a long record of coastal and marine biodiversity dating back at least two centuries. In terms of the marine environment, India has a coastline of about 8000 km including offshore islands and a very wide range of coastal ecosystems, such as estuaries, lagoons, mangroves, backwaters, salt marshes, rocky coasts, sandy beaches and coral reefs (Venkataraman and Wafar, 2005).
Goa, the smallest state of India, is located on the western coast of the Indian peninsula between the Arabian Sea and the mountain range of the Western Ghats. It shares its borders with Maharashtra to the north and Karnataka to the south. Most of Goa forms part of the coastal region known as the Konkan. Goa experiences tropical weather for most of the year; temperatures do not vary greatly and range between 19 and 32°C. Summer is hottest in May while the winter months of January and February are pleasantly cool. The southwest monsoon hits the state between the months of June and September, and July receives the highest rainfall (98 mm) while February is the driest month (Mascarenhas, 1999).
Accelerated loss of coastal and marine biodiversity components over the last few decades has been of great concern, with environmental changes, overexploitation and habitat loss among the major causes of species loss. Marine molluscs have been shown to be an appropriate indicator group for local invertebrate biodiversity. They can be used to test the suitability of methods for rapid biodiversity assessment because they encompass the full range of survival strategies, including a variety of dispersal mechanisms (Smith, 2005). Molluscs have been favoured in previous studies on rocky shores and intertidal zones because they are numerous, relatively slow moving and easy to identify. They form an important component of the intertidal biota, with a unique combination of traits that make them easy to study. First, molluscan shells persist relatively intact for some time after the death of the animal, increasing the chances of sampling seasonal and rare species. Second, species identification is usually possible at any growth stage. And third, they are readily visible with the naked eye (Benkendorff and Przeslawski, 2008).
Of the 80,000 to 100,000 species of molluscs recorded from various parts of the world, 3271 species are known to occur in India belonging to 220 families and 591 genera. These comprise 1900 gastropods, 1100 bivalves, 210 cephalopods, 41 polyplacophorans and 20 scaphopods (Venkataraman and Wafar, 2005). However, there is virtually no information available on the current status of the molluscan fauna along the Karnataka-Goa coastline, which forms a major geographical belt of the Western Ghats. The intertidal and sub-tidal zones are known to provide habitats for marine fauna whose patterns are subject to seasonal changes (Defeo et al., 2009). Therefore, the present study was undertaken to (1) prepare a checklist of the gastropod species that inhabit the intertidal and subtidal zones of rocky and sandy beaches in Goa, (2) measure species diversity and richness using diversity indices, and (3) estimate seasonal variation of gastropod diversity.
Study area, sampling procedure and data analysis Intertidal and subtidal zones were included in both regions along a coastline covering a distance of 100 km. Of the 16 selected sampling sites (figure 1), eight were located in north Goa (Vagator, Anjuna, Baga, Calangute, Candolim, Sinquerim, Miramar and Dona Paula) and eight were in south Goa (Velsao, Majorda, Betalbatim, Colva, Vacra, Betul, Palolem and Galjibaga). The sampling sites comprised six rocky shores (figure 2) (Sinquerim, Candolim and Anjuna in north Goa, and Vacra, Betul and Palolem in south Goa) and the remaining ten sites were sandy beaches. When selecting the sampling sites, their proximity to industry, harbours and human habitation was considered. Sites more than 30 km away from urban areas were also included in this investigation in order to ensure the representation of gastropod fauna in a range of habitats.
Surveys were conducted in the intertidal and subtidal zones during the pre-monsoon and post-monsoon seasons at all 16 sampling sites. The surveys were undertaken at low tide in clear weather and relatively calm sea conditions between January 2008 and September 2009. Each site was visited once in January, February, March and April during the pre-monsoon season and once again in June, July, August and September during the post-monsoon season. A timed 2-4 h search was conducted during each visit and a species list of molluscs was generated for each site. Molluscan species were collected (handpicked by the author with the assistance of local fishermen) across the entire range of habitats at all levels of the shore. Particular attention was paid to the undersides of boulders (figure 3), vertical rock faces, caves and crevices, and tidal pools on rock platforms. In all surveys live specimens were not disturbed but their images were captured for identification using a Nikon D60 digital SLR camera.
The collected shells were transported in large plastic bags that were numbered sequentially with a code name for each sampling site. Soon after arrival in the laboratory, the shells were soaked for 30 minutes in warm water mixed with a mild detergent to get rid of any mucus, debris and odour. They were then rinsed several times in tap water, dried in the shade for a week, and sorted and stored in plastic sachets. The shells were identified according to keys of Apte (1998) and Dance (2002). Where the names of species differed, the nomenclature of Dance (2002) was used.
Biodiversity can be quantified in many different ways. The two main factors taken into account when measuring diversity are species richness and evenness. The following biodiversity indices were calculated: the Shannon-Wiener index to characterise gastropod diversity; the Margalef index to estimate species richness; and Pielou’s index of non-randomness to determine species evenness. Data were then subjected to statistical analysis using SPSS version 13.0 software to compare mean diversity indices and seasonal variation for each individual species (Zar, 1984; Forthhofer et al., 2007).
Spatial distribution and gastropod diversity An overall total of 2493 individuals representing 85 species, 51 genera and 24 families was recorded from the 16 sampling sites in northern and southern regions Goa (Table 1). Cumulative records revealed that 51 species occurred exclusively in the intertidal zone of rocky shores, whereas 34 species were found in the subtidal zone of sandy shores and of these 15 species occurred in both zones. The numbers of both species and individuals were highest at Vacra (63 and 218, respectively) and lowest at Miramar (13 and 74).
Most individual shells represented just two families: Turritellidae and Trochidae.
During the pre-monsoon months, the Shannon-Wiener diversity index ranged from 2.1749 (Miramar, a sandy beach = S) to 5.436 (Betul, a rocky beach = R). The Margalef index of species richness was highest in Vacra (R) and lowest in Velsao (S), while Peilou’s evenness index was least at Sinquerim (R) and highest at Betalbatim (S) and Betul (R). In the post-monsoon season, the Shannon- Wiener values ranged from 2.558 (Dona Paula, S) to 5.7653 (Majorda, S), Margalef’s index was highest in Vacra (R) and lowest in Velsao (S), and Dona Paula (S), Velsao (S), Colva (S) and Majorda (S) showed maximum evenness.
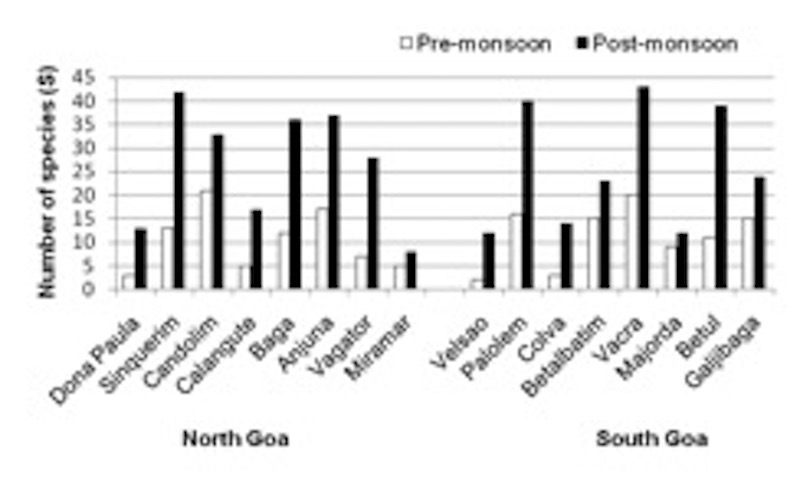
Seasonal variation The numbers of species collected at each site during the pre- and post-monsoon seasons are shown in figure 4. In the pre-monsoon season, the number of species recorded at five sites was less than or equal to five with maxima of 20 and 21 at two sites. After the monsoon season, the number of species recorded at all the sites increased and only five sites had fewer than 15 species, six sites had more than 35 and two had more than 40.
A significant (P<0.01) increase was observed in the total number of individuals of all species during the post-monsoon months compared to the pre-monsoon season. Monthly data from January to April showed a gradual decrease (P<0.01) in the total number of individuals during the four pre-monsoon months, and a significant (P<0.01) increase was observed in June and July, the first two months of the post-monsoon season followed by a significant (P<0.01) decrease in the total number of individuals in August and September.
Discussion The study revealed that more than 80 species of gastropods inhabit the coastal belt of Goa, India. Species richness was greatest in rocky intertidal or littoral zones, as reported for similar zones in south-eastern Australia (Benkendorff and Davis, 2002). The richest sites were Vacra, Sinquerim, Palolem and Candolim during the pre- and post-monsoon seasons. This may be due to the rough terrain of these sites where steep cliffs and creeks prevent anthropogenic disturbance. Subtidal zones of all the sandy beaches were low both in species richness and total number of individuals, owing to the extensive exploitation of these beaches for tourism. Dona Paula and Velsao showed especially poor species richness and low numbers of individuals since these two sites lie close to Mormugao harbor, where large amounts of toxic waste materials from shipping activities are released (Sarkar et al., 2008).
A striking result of this investigation was the significant increase in the total number of species and individuals in the post-monsoon season. The reason for this variation, whether related to temperature change or other environmental factors, remains to be studied.
The coastal zone of Goa has traditionally been used for agriculture, farming, shell fishing, fishing and low key recreation (Mascarenhas, 1999). The rapid increase in urbanisation and tourism appears to have adversely affected the biodiversity and distribution of the gastropod fauna in this region.
Acknowledgements The author is grateful to Dr Martin Punith and Ms Monisha Punith who assisted in fieldwork and the careful preservation of the specimens collected. The suggestions by Ms Veena Ammanna on data analysis are gratefully acknowledged. This work was supported by University Grants Commission, New Delhi, Minor Research Project, under the XI Plan, No. MRP (S)-222/08-09/KAKA063/UGC-SWRO.
References Apte, D. (1998). The book of Indian shells. Oxford University Press, Mumbai, India. Benkendorff, K. and Davis, A.R. (2002). Identifying hotspots of molluscan species richness on rocky intertidal reefs. Biodiversity and Conservation, 11: 1959–1973. Benkendorff, K. and Przeslawski, R. (2008). Multiple measures are necessary to assess rarity in macro-molluscs: a case study from southeastern Australia. Biodiversity and Conservation, 17: 2455–2478. Dance, S.P. (2002). Shells. Dorling Kindersley Ltd, London, UK. Defeo, O., McLachlan, A., Schoeman, D.S. et. al., (2009). Threats to sandy beach ecosystems: a review. Estuarine Coastal and Shelf Science, 81: 1-12. Forthforer, R.N., Lee, E. S. and Hernandez, M. (2007). Biostatistics: a guide to design, analysis and discovery. Academic Press, Elsevier Inc., California, USA. Mascarenhas, A. (1999). Some observations on the state of the coastal environment of Goa, west coast of India. Proceedings of the Integrated Coastal and Marine Area Management Plan for Goa, Department of Ocean Development, Chennai, pp. 204-224. Sarkar, A., Gaitonde, D. C. S., Sarkar, A., Vashistha, D., D’Silva, D. and Dalal, S.G. (2008). Evaluation of impairment of DNA integrity in marine gastropods (Cronia contracta) as a biomarker of genotoxic contaminants in coastal water around Goa, west coast of India. Ecotoxicology and Environment Safety, 71: 473–482. Smith, S. D. A. (2005). Rapid assessment of invertebrate biodiversity on rocky shores: where there’s a whelk there’s a way. Biodiversity and Conservation, 14: 3565–3576. Venkataraman, K. and Wafar, M. V. M. (2005). Coastal and marine biodiversity of India. Indian Journal of Marine Sciences, 34 (1): 57-75. Zar, J. H. (1984). Biostatistical analysis. 2nd edn. Prentice-Hall of Australia Pty. Ltd, Sydney, Australia. |
figure 1: Map showing approximate location of sampling sites along the coast of Goa. Rocky shores are underlined. (mapping: Peter Topley)
figure 2: Intertidal zone of a typical rocky shore in Goa. (Photo: Anuradha David)
figure 3: Live Turbo (Marmarostoma) bruneus (Photo: Anuradha David)
figure 4: Seasonal variation in species occurrence
|
The gastropod fauna of the Goa coastline
Issue
27
Page
21